
Through real-time fluorescent qPCR and next-generation sequencing (NGS) technologies in combination with data platforms, Luye Diagnostics provides clinical, intelligent, complete solutions in different stages of the disease.


Through real-time fluorescent qPCR and next-generation sequencing (NGS) technologies in combination with data platforms, Luye Diagnostics provides clinical, intelligent, complete solutions in different stages of the disease.
Vela Diagnostics, a member of Luye Diagnostics, is a world leading provider of integrated molecular solutions for the diagnosis. Vela pioneer in offering both qPCR and NGS, there’re over 30 CE-IVD and TGA tests including the first NGS test for HIV drug resistance.
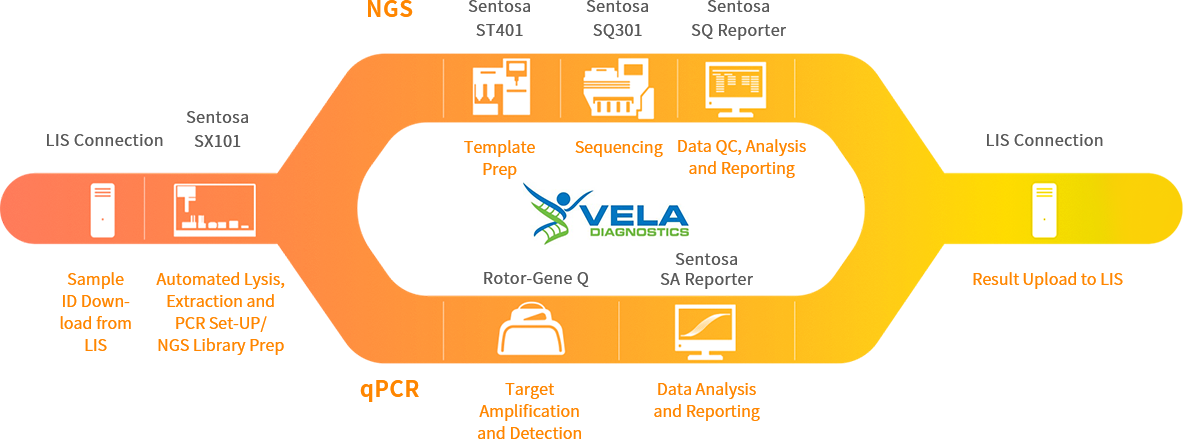
Fully automated NGS workflows from nucleic acid extraction, library prep, sequencing to bioinformatics analysis and report generation with simple, standardized and reliable results
Real-time fluorescent qPCR/POCT: Convenient, efficient and accurate molecular testing
Real-time fluorescent qPCR
Next-generation sequencing (NGS)
| Pathogen related diseases | Virus typing and drug resistance testing for infectious diseases: Multiple reports per test, 48 hours of TAT, and automated results reporting |
Testing of respiratory & intestinal pathogens, transplantation-related viruses, etc. Fast and wide coverage, providing help for the accurate diagnosis and treatment of pathogen-related diseases |
CMV, EBV, BKV,
HHV6, HSV-1/2
VZV
ISP FLEX
Influenza A/B & RSV
Flu/RSV
SARS-CoV-2
Direct MRSA/SA
HSV-1/2 Qual
HBV Quant
HCV Quant
HIV GT
HCV GT
HIV/HCV/HBV
CMV FLEX
Noro
| Tumour | Digital pathology platform: Providing systematic and standardized pathological testing and analysis services by integrating pathological image scanning, cloud data storage and AI analysis |
72 genetic mutations linked to breast cancer: A molecular diagnostic system suitable for the molecular typing, prognosis and immunity evaluation of Chinese breast cancer patients |
NSCLC
Breast cancer
Colon cancer
Thyroid cancer Melanoma
BCR-ABL (M)Qt
BCR-ABL (m)Qt
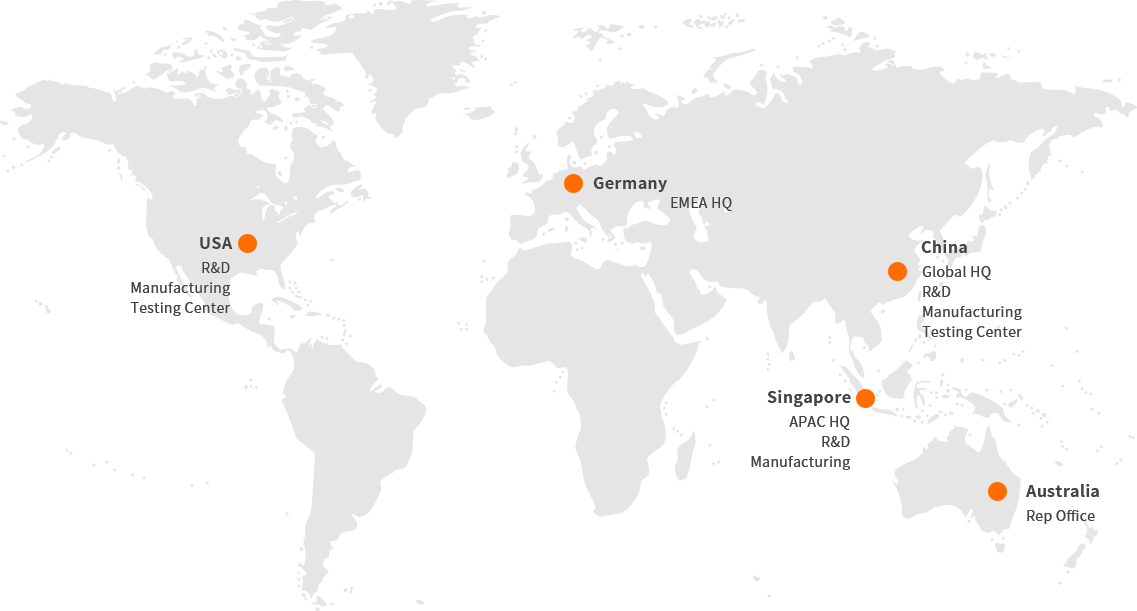

Operation, service and support networks across North America, Europe, and APAC

Regulatory approval in the United States, Europe, the Middle East, Asia Pacific and Africa, which facilitates rapid expansion. The Chinese market is expected to grow further in the next few years.

ViroKey™ SARS-CoV-2 RT-PCR Test obtained the EUA from the U.S. FDA, CE mark and provisional authorization from HSA for COVID-19 testing.
